Accelerating Class III Device Approval through Validated In Silico Evidence
Providing the high-fidelity computational modeling and ASME V&V 40 compliance required for BCI, Implantables, and Wearable safety validation.
- In Silico V&V
- HPC Infrastructure
- ASME V&V 40 Compliance
Validated Regulatory Evidence
Mehor Solutions provides high-fidelity electromagnetic modeling and validated In Silico evidence for mission-critical MedTech. From active implantables to complex wearables, we bridge the gap between initial R&D and formal FDA submission through rigorous verification frameworks and specialized regulatory evidence.
Clinical Application Expertise
Specialized electromagnetic verification for active medical devices, ensuring safety and performance compliance in the most complex human environments.
In Silico Design Verification
Utilizing a dedicated 2TB RAM HPC Cluster, we perform risk-informed computational modeling to confirm your device meets all electromagnetic specifications and safety margins before physical prototyping.

Regulatory Compliance (ASME V&V 40)
Our simulation workflows adhere to the ASME V&V 40 standard. We generate the validated technical artifacts, sensitivity studies, and worst-case analysis required for formal regulatory submissions.
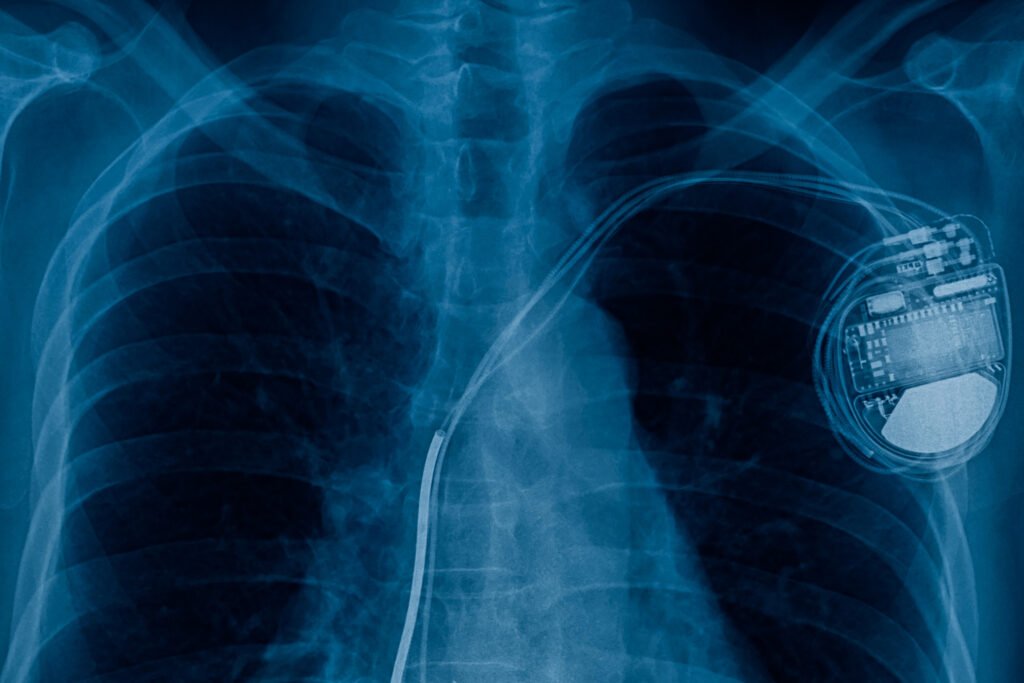
Implantables Systems
High-fidelity In Silico V&V for Class III devices: We provide the validated computational evidence required for ASME V&V 40 compliance, ensuring electromagnetic safety, link reliability, and SAR thermal safety in deep-tissue environments.
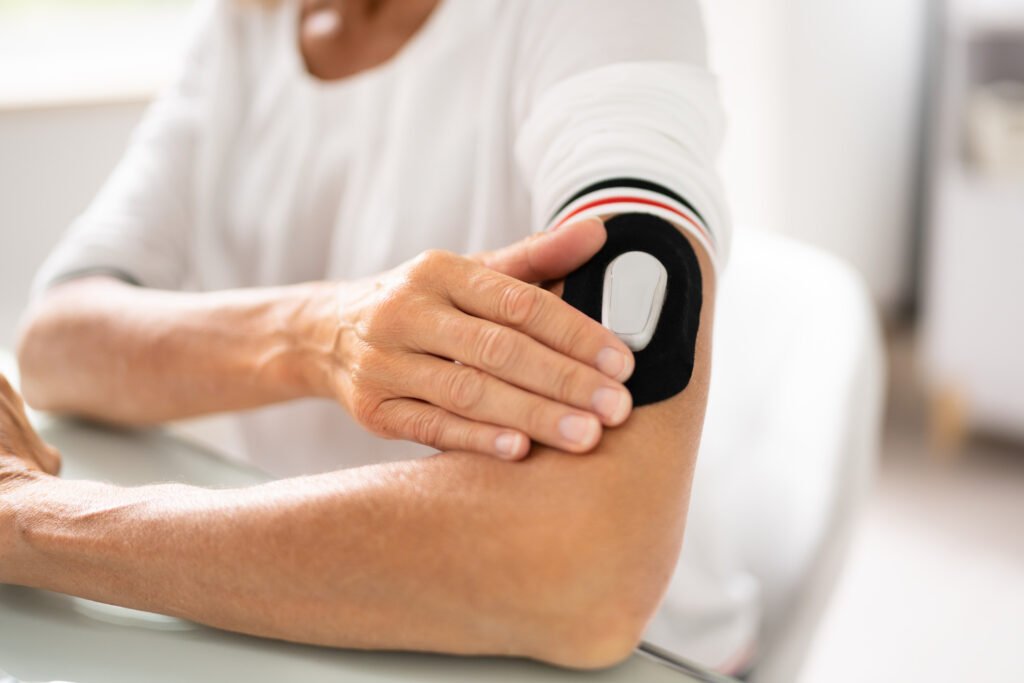
Wearable Technology
Precision modeling for skin-worn patches and diagnostic tools. We optimize impedance matching using advanced Voxel phantoms, confirming safety compliance and accelerating the move to regulatory submission while ensuring usability.
Collaborated with Industry Leaders & Innovative Startups
OSCOR • CAPRI MEDICAL • SYNKOPI • CURONIX
VALENTIUM • RF EXPOSURE LAB
ARIZONA STATE UNIVERSITY
Engagement Workflow

Step 1:
Initial Assessment
Submit a brief regarding your device type and regulatory timeline. We will conduct a preliminary review to ensure alignment with our technical capabilities.

Step 2:
Technical Strategy Session
A deep-dive consultation between our Principal Engineer and your technical leads. We establish an NDA and define the specific In Silico evidence required for your project.

Step 3:
Scope & V&V Framework
We provide a detailed proposal outlining the computational resources (HPC), modeling methodology, and the ASME V&V 40 framework to be applied.

Step 4:
Project Initiation
Upon agreement of terms, we integrate with your R&D timeline to begin generating the validated electromagnetic evidence needed for your submission.
